Netflix is often held up as a classic example of disruption. Netflix put Blockbuster out of business because it offered consumers a less expensive way to rent movies without the inconvenience of traveling to Blockbuster stores. Describing Netflix as “disruptive” is only half the story. “To whom” Netflix was disruptive is the other half. Netflix disrupted Blockbuster.
At the Disruptive Innovations to Advance Clinical Trials Conference in Boston on September 19-20, 2013, dozens of companies positioned themselves as “disruptive,” but few offered the other half of the story. The most important question should have been, “To whom are these companies disruptive (if at all)?” By answering this question, we can gain a clear understanding of the future of the industry so we can “skate to where the puck will be,” rather than just investing incrementally in where it is today.
Some of the key themes from the conference included the role of physicians / sites in trials, how disruptive technologies are changing this role, and what the theory of disruptive innovation predicts for the future of clinical trials.
Types of Innovation in Clinical Trials
Glen de Vries, the President of Medidata Solutions, (full disclosure: I work for Medidata as Director of Strategy*) referenced “the Clay Christensen definition of disruptive innovation” as taking activities that have traditionally been done by highly trained professionals in high cost venues and making them simple, affordable, and flexible enough so that they could be done by less skilled people in more convenient, lower cost settings. By contrast, an innovation that helps these highly trained professionals do their job more efficiently or in a more sophisticated way is not actually “disruptive.” It would be “sustaining” to the established ecosystem.
The job-to-be-done of a clinical trial is to help life sciences companies obtain approval for their new therapies. This includes designing trials, recruiting and enrolling the right patients, delivering therapies to patients and making sure they take them, monitoring patient progress, ensuring compliance to the plan, analyzing the results, and submitting them to regulators for approval.
As long as the job of a clinical trial remains constant, the need for these activities will also remain constant. However, the ways of accomplishing them may change.
Optimizing Site Activities is Sustaining to the Site Model
Life science companies have traditionally hired sites (i.e., doctors) to recruit patients, deliver therapies, and monitor patient progress. Many companies at the conference called themselves “disruptive,” but they are actually “sustaining” to this site-centric model. They help sites do their jobs better and more efficiently, while improving the profitability of life sciences companies. For example:
- Site selection tools (ViS Research, iTrials, and Investigator DataBank consortium)
- Risk-based monitoring, which reduces the cost and burden of site management through more targeted oversight (Eugene Patin from Novartis)
- Electronic patient pre-registration, screening, and consent to make patient on-boarding more efficient at sites (eConsent, Joseph Kim from Shire)
- Electronic investigator payments (Greenphire)
- Site training and credentialing (Dalvir Gill from the TransCelerate consortium)
While these are sustaining to the site model, they have the potential to be disruptive in their own ways. eConsent and Greenphire, for example, are a simpler and more affordable alternative to paper technologies and may help attract less sophisticated, non-participating sites to start conducting clinical trials. They may also be disruptive to CROs by making it simpler and more affordable for sponsors to manage their own trials.
Moving Activities Away from Sites is Disruptive to the Site Model
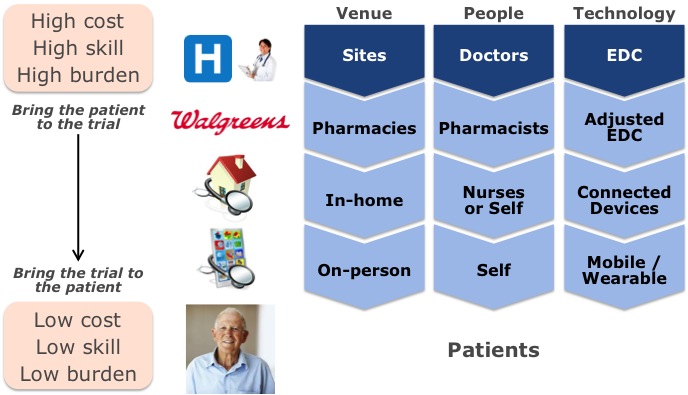
Several people discussed ways to disrupt the site model by making the clinical trials process more “patient centric.” Only the patients who are most altruistic, in the greatest medical need, or who coincidentally live near investigative sites participate. Clinical trials could become more accessible to patients by moving clinical trial activities away from high cost, high skill, and inconvenient settings and into lower cost, lower skill, and more convenient settings. As technology matures in these new settings, clinical trial activities will migrate to them.
1. Pharmacies
Walgreens and Novartis presented a pilot program in which an entire clinical trial was run using pharmacies alone. In a separate presentation, Komathi Stern (Genentech/Roche) said only 30% of patients live near current investigative sites, but 80% of patients would join a trial if the site were within 30 minutes of their home. Over 67% of the U.S. population lives within 3 miles of a Walgreens, and 40% of the population lives in medically underserved communities. Therefore, the more distributed infrastructure of pharmacies may provide patients greater accessibility to trials than investigator sites. Walgreens’ comprehensive patient database, low-cost point-of-care diagnostics, and network of visiting nurses could help recruit patients and manage trials either in-store or at-home. In addition to greater access to patients, the cost of providing care in pharmacies or via a network of visiting nurses is estimated to be up to 50% lower than at a doctor’s office.
2. In-home/Mobile Patient Monitoring
Medidata Solutions and Spaulding Clinical Research were awarded the 2013 DPharm Disruptive Innovator of the Year Award at the conference for incorporating connected mHealth devices into their own clinical trial. Traditionally, patient data has been recorded at sites using Electronic Data Capture (EDC) tools. As the locus of trials shifts closer to the patient, the technologies to capture patient data for trials need to move as well. With its roots in EDC, Medidata is expanding its cloud platform to integrate data from connected medical and mHealth devices to form a holistic view of a patient over time. Spaulding’s portable ECG machine, as well as a Withings fitness tracker and scale, represents this foray into remote patient monitoring. This approach to data capture is much less burdensome to patients, improving their likelihood to participate and remain engaged in trials. It also can capture more frequent or continuous data points and in a less expensive way, providing new insights and potentially higher data quality while reducing the cost of trials.
3. Online Patient Recruiting
Jeff Kingsley from SERRG described today’s paradigm: “We need sites because that’s where patients come from.” Yet many presentations identified online health-oriented sites like PatientsLikeMe, WebMD, and Patient Advocacy Groups as viable alternatives for patient recruitment in the future. Bernhard Warner from Bloomberg Business Week suggested that mobile technologies could help recruit and manage patients in emerging countries, where site infrastructure is less developed, particularly for rare disease trials.
4. Telemedicine
Telemedicine can be used to support virtual visits, as discussed by Craig Lipset (Pfizer), Marc Foster (Transparency Life Sciences), and Komathi Stern (Genentech/Roche).
The Road Ahead
There is tremendous opportunity for both entrepreneurs and established companies to help build out this disruptive value network. In a space crowded with EDC competitors supporting the site-based model of data capture, entrepreneurs and larger companies alike should “skate to where the puck will be” by developing low cost, portable monitoring sensors for use in-pharmacies, in-home, on-body, and eventually in-body. Several presentations at the conference alluded to the likelihood that a facilitated network platform will integrate this decentralized ecosystem of devices, apps, and supporting services into the trials process.
Similarly, life sciences companies need to embrace this emerging value network. These new approaches for running trials are sustaining to them, for they have the potential to dramatically lower development costs, reduce time to market, mitigate risk, and increase the probability of trial success. Process change is hard, but embracing disruptive change is essential to remaining competitive over the longer term.
Netflix offered consumers more affordable access to movies in home and later on mobile devices. Disruption in clinical trials is moving in the same direction, and as the era of Blockbuster drugs fades (pun intended), we need to be prepared for what’s next.
*There is no financial relationship between Medidata and the Christensen Institute.